|
12/8/2023 0 Comments Spike protein in ovaries( C) Size-Exclusion HPLC chromatogram of purified SΔTM (shown as blue line) and a 670 kDa molecular weight standard (shown as black line). Molecular weight standards are indicated at the left in kDa. The structure model of S-trimer was generated by the SWISS-MODEL using homology modelling techniques ( ), and the 3D structure figures were prepared using PyMOL ( ( B) SDS-PAGE analysis of purified SΔTM and RBD. Two recombinant SARS-CoV-2 spike antigens were designed: SΔTM (the prefusion S ectodomain with proline substitutions at residues 986 and 987 to retain S2 in the prefusion conformation, a “GGSG” substitution at the furin cleavage site, a C-terminal T4 fibritin trimerization motif), and 260-mer RBD (RBD-SD1). SS, signal sequence NTD, N-terminal domain RBD, receptor-binding domain SD1, subdomain 1 SD2, subdomain 2 S1/S2, S1/S2 protease cleavage site S2′, S2′ protease cleavage site FP, fusion peptide HR1, heptad repeat 1 CH, central helix CD, connector domain HR2, heptad repeat 2 TM, transmembrane domain CT, cytoplasmic tail. ( A) Domain architecture of the SARS-CoV-2 S protein. Molecular design and characterization of SΔTM and RBD. SARS-CoV-2 Subunit vaccine Trimeric spike protein.Ĭopyright © 2021. These data suggest candidate vaccine is efficacious in preventing SARS-CoV-2 infections and associated pneumonia, thereby justifying ongoing phase I/II clinical studies in China ( NCT04982068 and NCT04990544). In addition, the vaccine-induced antibodies showed cross-neutralization activity against B.1.1.7 and B.1.351 variants. Challenge studies demonstrated that candidate vaccine reduced the viral loads and inflammation in the lungs of SARS-CoV-2 infected golden Syrian hamsters significantly. And vaccine-induced neutralizing antibodies persisted at high level for at least 6 months. Immunogenicity studies showed that candidate vaccines elicited robust neutralizing antibody responses and substantial CD4 + T cell responses in both mice and non-human primates. 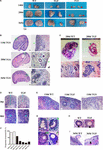
The spike protein was expressed in Chinese hamster ovary (CHO) cells, purified, and prepared as a stable formulation with the dual adjuvant. We have been developing a recombinant vaccine based on a prefusion-stabilized spike trimer of SARS-CoV-2 and formulated with aluminium hydroxide and CpG 7909. A safe, effective, and affordable vaccine against SARS-CoV-2 infections/diseases is urgently needed. Electronic address: pandemic has severely impacted the public health and social economy worldwide. Electronic address: 9 Shanghai Zerun Biotechnology Co., Ltd., Shanghai, China. Electronic address: 8 Institute of Laboratory Animal Sciences, Chinese Academy of Medical Sciences and Comparative Medicine Center, Peking Union Medical College, Beijing, China. Electronic address: 7 Key Laboratory of Medical Molecular Virology (MOE/NHC/CAMS), Department of Medical Microbiology and Parasitology, School of Basic Medical Sciences, Shanghai Medical College, Fudan University, Shanghai, China. 6 National Institute for Food and Drug Control (NIFDC), Beijing, China. 
5 State Key Laboratory of Virology, Modern Virology Research Center, College of Life Sciences, Wuhan University, Wuhan, China.4 Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, China.3 Key Laboratory of Medical Molecular Virology (MOE/NHC/CAMS), Department of Medical Microbiology and Parasitology, School of Basic Medical Sciences, Shanghai Medical College, Fudan University, Shanghai, China. 
2 Institute of Laboratory Animal Sciences, Chinese Academy of Medical Sciences and Comparative Medicine Center, Peking Union Medical College, Beijing, China.1 Shanghai Zerun Biotechnology Co., Ltd., Shanghai, China.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
